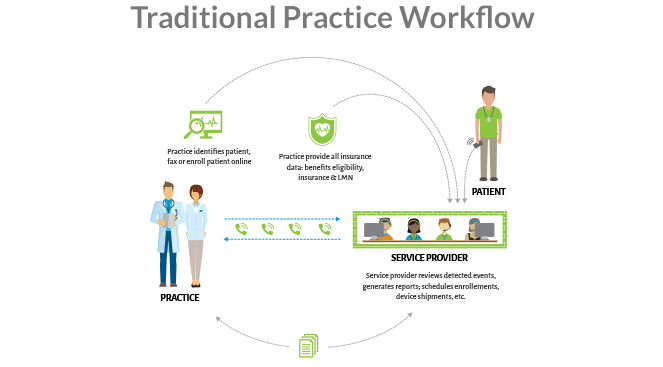
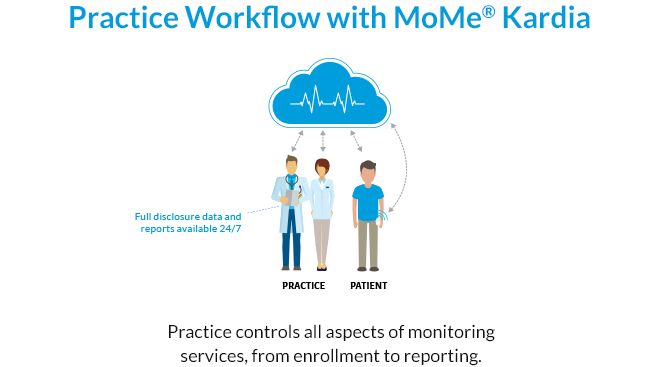
MoMe® Kardia delivers on the full promise of full disclosure. See How at HRS 2019.
Demo MoMe® Kardia at BIOTRONIK Booth #219 and learn how you can gain a virtual telemetry station for MCT monitoring, anywhere.

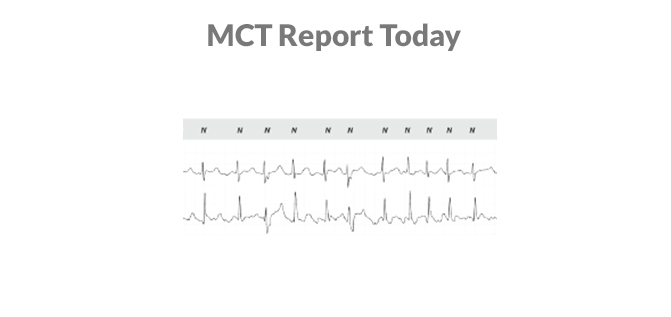
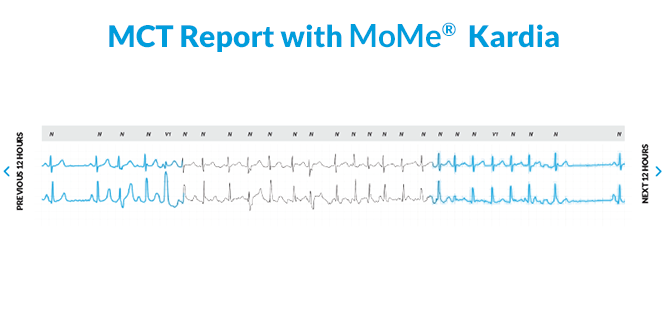
Yes, true MCT does exist. See it for yourself at HRS Booth #219. We’ll show you why MoMe® Kardia is the only system that brings the 24/7 data and compliance benefits of inpatient telemetry to ambulatory cardiac monitoring.